| Entry |
|
|---|
| Name |
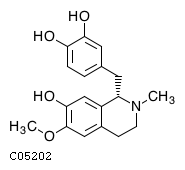
3'-Hydroxy-N-methyl-(S)-coclaurine;
(S)-3'-Hydroxy-N-methylcoclaurine
|
|---|
| Formula |
C18H21NO4
|
|---|
| Exact mass |
315.1471
|
|---|
| Mol weight |
315.36
|
|---|
| Structure |
|
|---|
| Reaction |
|
|---|
| Pathway |
| map00950 | Isoquinoline alkaloid biosynthesis |
| map01063 | Biosynthesis of alkaloids derived from shikimate pathway |
| map01110 | Biosynthesis of secondary metabolites |
|
|---|
| Module |
| M00943 | Reticuline biosynthesis, dopamine + 4HPAA => (S)-reticuline |
|
|---|
| Enzyme |
|
|---|
| Brite |
Phytochemical compounds [BR:br08003]
Alkaloids
Alkaloids derived from tyrosine
Isoquinoline alkaloids
C05202 3'-Hydroxy-N-methyl-(S)-coclaurine
|
|---|
| Other DBs |
|
|---|
| LinkDB |
|
|---|
| KCF data |
ATOM 23
1 C8y C 27.0948 -22.6553
2 C1y C 28.3008 -21.9562
3 C8y C 27.0890 -24.0536
4 C8x C 25.8830 -21.9503
5 C1b C 28.3065 -20.5520
6 N1y N 29.5125 -22.6611
7 C8x C 25.8830 -24.7528
8 C1x C 28.2891 -24.7585
9 C8y C 24.6654 -22.6553
10 C8y C 27.1064 -19.8530
11 C1x C 29.5068 -24.0653
12 C1a C 30.7302 -21.9619
13 C8y C 24.6654 -24.0536
14 O1a O 23.4534 -21.9503
15 C8x C 27.1005 -18.4488
16 C8x C 25.9004 -20.5520
17 O2a O 23.4534 -24.7528
18 C8y C 25.8830 -17.7497
19 C8x C 24.6828 -19.8645
20 C1a C 22.2358 -24.0536
21 C8y C 24.6770 -18.4605
22 O1a O 25.8771 -16.3573
23 O1a O 23.4594 -17.7614
BOND 25
1 1 2 1
2 1 3 1
3 1 4 2
4 2 5 1 #Down
5 2 6 1
6 3 7 2
7 3 8 1
8 4 9 1
9 5 10 1
10 6 11 1
11 6 12 1
12 7 13 1
13 9 14 1
14 10 15 1
15 10 16 2
16 13 17 1
17 15 18 2
18 16 19 1
19 17 20 1
20 18 21 1
21 18 22 1
22 21 23 1
23 8 11 1
24 9 13 2
25 19 21 2
|
|---|