 | | DRUG: Ibuprofen | |
| Entry |
|
|---|
| Name |
Ibuprofen (JP18/USP/INN);
Advil (TN);
Motrin (TN) |
|---|
| Product |
|
|---|
| Generic |
IBU (Aphena Pharma Solutions - Tennessee), IBU (Aphena Pharma Solutions - Tennessee), IBU (Aphena Pharma Solutions - Tennessee), IBU (Dr. Reddy's Laboratories Limited), IBU (Golden State Medical Supply), IBU (Lake Erie Medical DBA Quality Care Products LLC), IBU (NuCare Pharmaceuticals), IBU (NuCare Pharmaceuticals), IBU (NuCare Pharmaceuticals), IBU (Proficient Rx LP), IBU (Proficient Rx LP), IBU (RedPharm Drug), IBU (RedPharm Drug), IBU (RedPharm Drug), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (A-S Medication Solutions), IBUPROFEN (Actavis Pharma), IBUPROFEN (Advanced Rx Pharmacy of Tennessee), IBUPROFEN (Advanced Rx Pharmacy of Tennessee), IBUPROFEN (Advanced Rx Pharmacy of Tennessee), IBUPROFEN (Advanced Rx of Tennessee), IBUPROFEN (Advanced Rx of Tennessee), IBUPROFEN (Advanced Rx of Tennessee), IBUPROFEN (Advanced Rx of Tennessee), IBUPROFEN (Advanced Rx of Tennessee), IBUPROFEN (Advanced Rx of Tennessee), IBUPROFEN (American Health Packaging), IBUPROFEN (Amneal Pharmaceuticals LLC), IBUPROFEN (Amneal Pharmaceuticals NY LLC), IBUPROFEN (Amneal Pharmaceuticals of New York LLC), IBUPROFEN (Amneal Pharmaceuticals of New York LLC), IBUPROFEN (Amneal Pharmaceuticals of New York LLC), IBUPROFEN (Amneal Pharmaceuticals of New York LLC), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Aphena Pharma Solutions - Tennessee), IBUPROFEN (Ascend Laboratories), IBUPROFEN (Asclemed USA), IBUPROFEN (Asclemed USA), IBUPROFEN (Asclemed USA), IBUPROFEN (Aurobindo Pharma Limited), IBUPROFEN (Aurobindo Pharma Limited), IBUPROFEN (Brisk Pharmaceuticals), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Bryant Ranch Prepack), IBUPROFEN (Cardinal Health 107), IBUPROFEN (Chartwell RX), IBUPROFEN (Coupler LLC), IBUPROFEN (Coupler LLC), IBUPROFEN (Coupler LLC), IBUPROFEN (DIRECT RX), IBUPROFEN (Direct Rx), IBUPROFEN (Direct_Rx), IBUPROFEN (Direct_Rx), IBUPROFEN (Direct_rx), IBUPROFEN (EXELAN PHARMACEUTICALS), IBUPROFEN (Florida Pharmaceutical Products), IBUPROFEN (Golden State Medical Supply), IBUPROFEN (Granules India Limited), IBUPROFEN (Kesin Pharma Corporation), IBUPROFEN (Legacy Pharmaceutical Packaging), IBUPROFEN (MARKSANS PHARMA LIMITED), IBUPROFEN (Major Pharmaceuticals), IBUPROFEN (NCS HealthCare of KY), IBUPROFEN (NCS HealthCare of KY), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (Northwind Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (NuCare Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PD-Rx Pharmaceuticals), IBUPROFEN (PHOENIX RX LLC), IBUPROFEN (PHOENIX RX LLC), IBUPROFEN (Padagis Israel Pharmaceuticals), IBUPROFEN (PharmPak), IBUPROFEN (PharmPak), IBUPROFEN (PharmPak), IBUPROFEN (Pharmasource Meds), IBUPROFEN (Praxis), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Preferred Pharmaceuticals), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (Proficient Rx LP), IBUPROFEN (QPharma), IBUPROFEN (QPharma), IBUPROFEN (Quality Care Products LLC), IBUPROFEN (Quality Care Products), IBUPROFEN (Quality Care Products), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (REMEDYREPACK), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (RedPharm Drug), IBUPROFEN (Redpharm Drug), IBUPROFEN (Redpharm Drug), IBUPROFEN (Redpharm Drug), IBUPROFEN (Redpharm Drug), IBUPROFEN (SKYA HEALTH), IBUPROFEN (SOLA Pharmaceuticals), IBUPROFEN (SOLA Pharmaceuticals), IBUPROFEN (ST. MARY'S MEDICAL PARK PHARMACY), IBUPROFEN (St. Mary's Medical Park Pharmacy), IBUPROFEN (St. Mary's Medical Park Pharmacy), IBUPROFEN (St. Mary's Medical Park Pharmacy), IBUPROFEN (St. Mary's Medical Park Pharmacy), IBUPROFEN (Strides Pharma Science Limited), IBUPROFEN (Strides Pharma Science Limited), IBUPROFEN (Strides Pharma), IBUPROFEN (Sun Pharmaceutical Industries), IBUPROFEN (Sun Pharmaceutical Industries), IBUPROFEN (TIME CAP LABORATORIES), IBUPROFEN (Westminster Pharmaceuticals) |
|---|
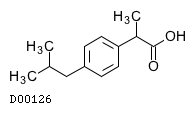
| Formula |
C13H18O2
|
|---|
| Exact mass |
206.1307
|
|---|
| Mol weight |
206.28
|
|---|
| Structure |
|
|---|
| Simcomp |
|
|---|
| Class |
Anti-inflammatory
DG01504 Nonsteroidal anti-inflammatory drug (NSAID)
DG01908 Anti-inflammatory drug, propionic acid derivative
Metabolizing enzyme substrate
DG01642 CYP2C9 substrate
|
|---|
| Remark |
| Therapeutic category: | 1149 |
| Product (DG00245): | D00126<JP/US> D01122<JP> D06606<JP/US> |
| Product (mixture): | D02152<US> D11575<US> D11897<US> |
|
|---|
| Efficacy |
Analgesic, Anti-inflammatory, Antipyretic, COX inhibitor |
|---|
| Comment |
Propionic acid derivative
|
|---|
| Target |
|
|---|
| Pathway |
|
|---|
| Metabolism |
Enzyme: CYP2C9 [HSA: 1559]
|
|---|
| Interaction |
|
|---|
| Structure map |
|
|---|
| Brite |
Anatomical Therapeutic Chemical (ATC) classification [BR:br08303]
C CARDIOVASCULAR SYSTEM
C01 CARDIAC THERAPY
C01E OTHER CARDIAC PREPARATIONS
C01EB Other cardiac preparations
C01EB16 Ibuprofen
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
G GENITO URINARY SYSTEM AND SEX HORMONES
G02 OTHER GYNECOLOGICALS
G02C OTHER GYNECOLOGICALS
G02CC Antiinflammatory products for vaginal administration
G02CC01 Ibuprofen
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
M MUSCULO-SKELETAL SYSTEM
M01 ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS
M01A ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS, NON-STEROIDS
M01AE Propionic acid derivatives
M01AE01 Ibuprofen
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
M02 TOPICAL PRODUCTS FOR JOINT AND MUSCULAR PAIN
M02A TOPICAL PRODUCTS FOR JOINT AND MUSCULAR PAIN
M02AA Antiinflammatory preparations, non-steroids for topical use
M02AA13 Ibuprofen
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
R RESPIRATORY SYSTEM
R02 THROAT PREPARATIONS
R02A THROAT PREPARATIONS
R02AX Other throat preparations
R02AX02 Ibuprofen
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
USP drug classification [BR:br08302]
Analgesics
Nonsteroidal Anti-inflammatory Drugs
Ibuprofen
D00126 Ibuprofen (JP18/USP/INN)
Therapeutic category of drugs in Japan [BR:br08301]
1 Agents affecting nervous system and sensory organs
11 Agents affecting central nervous system
114 Antipyretics and analgesics, anti-inflammatory agents
1149 Others
D00126 Ibuprofen (JP18/USP/INN)
Classification of Japanese OTC drugs [BR:br08313]
Agents for nervous and sensory systems
01 Cold remedy (oral use)
D00126 Ibuprofen (JP18/USP/INN)
03 Antipyretic analgesic agents
D00126 Ibuprofen (JP18/USP/INN)
Risk category of Japanese OTC drugs [BR:br08312]
Designated second-class OTC drugs
Inorganic and organic chemicals
Ibuprofen
D00126 Ibuprofen (JP18/USP/INN)
Second-class OTC drugs
Inorganic and organic chemicals
Ibuprofen
D00126 Ibuprofen (JP18/USP/INN)
Drug groups [BR:br08330]
Anti-inflammatory
DG01504 Nonsteroidal anti-inflammatory drug (NSAID)
DG01908 Anti-inflammatory drug, propionic acid derivative
DG00245 Ibuprofen
D00126 Ibuprofen
Metabolizing enzyme substrate
DG01642 CYP2C9 substrate
DG00245 Ibuprofen
D00126 Ibuprofen
Drug classes [BR:br08332]
Anti-inflammatory
DG01504 Nonsteroidal anti-inflammatory drug (NSAID)
D00126 Ibuprofen
Target-based classification of drugs [BR:br08310]
Enzymes
Oxidoreductases (EC1)
Prostaglandin synthase
PTGS1 (COX1)
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
PTGS2 (COX2)
D00126 Ibuprofen (JP18/USP/INN) <JP/US>
Drugs listed in the Japanese Pharmacopoeia [BR:br08311]
Chemicals
D00126 Ibuprofen
New drug approvals in Europe [br08329.html]
European public assessment reports (EPAR) authorised medicine
D00126
Drug metabolizing enzymes and transporters [br08309.html]
Drug metabolizing enzymes
D00126
Prodrugs [br08324.html]
D00126
Rx-to-OTC switch list in the USA [br08315.html]
D00126
Rx-to-OTC switch list in Japan [br08314.html]
D00126
Drug groups [BR:br08330]
Anti-inflammatory
DG01504 Nonsteroidal anti-inflammatory drug (NSAID)
DG01908 Anti-inflammatory drug, propionic acid derivative
DG00245 Ibuprofen
Metabolizing enzyme substrate
DG01642 CYP2C9 substrate
DG00245 Ibuprofen
|
|---|
| Other DBs |
|
|---|
| LinkDB |
|
|---|
| KCF data |
ATOM 15
1 O6a O 26.6164 -16.3136
2 C6a C 25.4050 -17.0138
3 C1c C 24.1928 -16.3148
4 O6a O 25.4056 -18.4130
5 C8y C 22.9815 -17.0150
6 C8x C 21.7750 -16.3132
7 C8x C 20.5638 -17.0135
8 C8y C 20.5644 -18.4127
9 C8x C 21.7765 -19.1116
10 C8x C 22.9878 -18.4113
11 C1b C 19.3530 -19.1128
12 C1c C 18.1408 -18.4139
13 C1a C 16.9295 -19.1141
14 C1a C 18.1401 -17.0147
15 C1a C 24.1920 -14.9103
BOND 15
1 2 4 2
2 3 5 1
3 5 6 2
4 6 7 1
5 7 8 2
6 8 9 1
7 9 10 2
8 10 5 1
9 8 11 1
10 1 2 1
11 11 12 1
12 12 13 1
13 2 3 1
14 12 14 1
15 3 15 1
|
|---|
|
» Japanese version » Back
|