 | | DRUG: ジクロフェナクナトリウム | |
| エントリ |
|
|---|
| 一般名 |
ジクロフェナクナトリウム (JP18);
Diclofenac sodium (JP18/USP) |
|---|
| 商品名 |
ジクトル (久光製薬), ジクロフェナクNa (三友薬品), ジクロフェナクNa (三友薬品), ジクロフェナクNa (日本臓器製薬), ジクロフェナクNa (日本臓器製薬), ジクロフェナクNa (東和薬品), ジクロフェナクナトリウム (ニプロファーマ), ジクロフェナクナトリウム (三和化学研究所), ジクロフェナクナトリウム (帝國製薬), ジクロフェナクナトリウム (祐徳薬品工業), ジクロード (わかもと製薬), ナボール (久光製薬), ナボール (久光製薬), ナボール (久光製薬), ナボール (久光製薬), ナボール (久光製薬), ボルタレン (ノバルティスファーマ), ボルタレン (ノバルティスファーマ), ボルタレン (同仁医薬化工), ボルタレン (同仁医薬化工), ボルタレン (同仁医薬化工), ボルタレン (同仁医薬化工) |
|---|
| 後発品 |
ジクロフェナクNa (ロートニッテン), ジクロフェナクNa (ロートニッテンファーマ), ジクロフェナクNa (ロートニッテンファーマ), ジクロフェナクNa (三友薬品), ジクロフェナクNa (日医工岐阜工場), ジクロフェナクNa (日医工岐阜工場), ジクロフェナクNa (日新製薬-山形), ジクロフェナクNa (日新製薬-山形), ジクロフェナクNa (日新製薬-山形), ジクロフェナクNa (日本臓器製薬), ジクロフェナクNa (東亜薬品), ジクロフェナクNa (東光薬品工業), ジクロフェナクNa (東光薬品工業), ジクロフェナクNa (東和薬品), ジクロフェナクNa (沢井製薬), ジクロフェナクNa (辰巳化学), ジクロフェナクNa (辰巳化学), ジクロフェナクNa (陽進堂), ジクロフェナクNa (鶴原製薬), ジクロフェナクナトリウム (京都薬品工業), ジクロフェナクナトリウム (帝國製薬), ジクロフェナクナトリウム (日医工), ジクロフェナクナトリウム (日本ジェネリック), ジクロフェナクナトリウム (祐徳薬品工業) |
|---|
| 米国の商品 |
|
|---|
| 後発品 |
DICLOFENAC (Advanced Rx of Tennessee), DICLOFENAC (Altaire Pharmaceuticals), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (A-S Medication Solutions), DICLOFENAC SODIUM (ASCLEMED USA), DICLOFENAC SODIUM (Actavis Pharma), DICLOFENAC SODIUM (Actavis Pharma), DICLOFENAC SODIUM (Advagen Pharma), DICLOFENAC SODIUM (Advagen Pharma), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx Pharmacy of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Advanced Rx of Tennessee), DICLOFENAC SODIUM (Alembic Pharmaceuticals Limited), DICLOFENAC SODIUM (Alembic Pharmaceuticals Limited), DICLOFENAC SODIUM (Alembic Pharmaceuticals), DICLOFENAC SODIUM (Alembic Pharmaceuticals), DICLOFENAC SODIUM (American Health Packaging), DICLOFENAC SODIUM (American Health Packaging), DICLOFENAC SODIUM (Amneal Pharmaceuticals LLC), DICLOFENAC SODIUM (Amneal Pharmaceuticals LLC), DICLOFENAC SODIUM (Amneal Pharmaceuticals of New York LLC), DICLOFENAC SODIUM (Aphena Pharma Solutions - Tennessee), DICLOFENAC SODIUM (Apotex Corp.), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Asclemed USA), DICLOFENAC SODIUM (Aurobindo Pharma Limited), DICLOFENAC SODIUM (Aurolife Pharma LLC), DICLOFENAC SODIUM (AvKARE), DICLOFENAC SODIUM (AvKARE), DICLOFENAC SODIUM (Bausch & Lomb Incorporated), DICLOFENAC SODIUM (BluePoint Laboratories), DICLOFENAC SODIUM (BluePoint Laboratories), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Bryant Ranch Prepack), DICLOFENAC SODIUM (Carlsbad Technology), DICLOFENAC SODIUM (Carlsbad Technology), DICLOFENAC SODIUM (Coupler LLC), DICLOFENAC SODIUM (DIRECT RX), DICLOFENAC SODIUM (DirectRx), DICLOFENAC SODIUM (Direct_Rx), DICLOFENAC SODIUM (Direct_Rx), DICLOFENAC SODIUM (Edenbridge Pharmaceuticals LLC.), DICLOFENAC SODIUM (Encube Ethicals), DICLOFENAC SODIUM (GLENMARK PHARMACEUTICALS), DICLOFENAC SODIUM (Gabar Health Sciences Corp), DICLOFENAC SODIUM (Golden State Medical Supply), DICLOFENAC SODIUM (Golden State Medical Supply), DICLOFENAC SODIUM (Ingenus Pharmaceuticals), DICLOFENAC SODIUM (Lannett Company), DICLOFENAC SODIUM (Lupin Pharmaceuticals), DICLOFENAC SODIUM (Lupin Pharmaceuticals), DICLOFENAC SODIUM (Medsource Pharmaceuticals), DICLOFENAC SODIUM (Medsource Pharmaceuticals), DICLOFENAC SODIUM (NorthStar Rx LLC), DICLOFENAC SODIUM (NorthStar Rx LLC), DICLOFENAC SODIUM (Northwind Pharmaceuticals), DICLOFENAC SODIUM (Northwind Pharmaceuticals), DICLOFENAC SODIUM (Novel Laboratories), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (NuCare Pharmaceuticals), DICLOFENAC SODIUM (Oceanside Pharmaceuticals), DICLOFENAC SODIUM (PD-Rx Pharmaceuticals), DICLOFENAC SODIUM (PD-Rx Pharmaceuticals), DICLOFENAC SODIUM (PHOENIX RX LLC), DICLOFENAC SODIUM (PHOENIX RX LLC), DICLOFENAC SODIUM (PHOENIX RX LLC), DICLOFENAC SODIUM (PHOENIX RX LLC), DICLOFENAC SODIUM (PHOENIX RX LLC), DICLOFENAC SODIUM (Padagis Israel Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Preferred Pharmaceuticals), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Proficient Rx LP), DICLOFENAC SODIUM (Quality Care Products LLC), DICLOFENAC SODIUM (Quality Care Products LLC), DICLOFENAC SODIUM (Quality Care Products), DICLOFENAC SODIUM (Quality Care Products), DICLOFENAC SODIUM (Quality Care Products), DICLOFENAC SODIUM (Quality Care Products), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REMEDYREPACK), DICLOFENAC SODIUM (REPHARM LLC), DICLOFENAC SODIUM (RedPharm Drug), DICLOFENAC SODIUM (RedPharm Drug), DICLOFENAC SODIUM (RedPharm Drug), DICLOFENAC SODIUM (RedPharm Drug), DICLOFENAC SODIUM (Redpharm Drug), DICLOFENAC SODIUM (Rising Pharma Holdings), DICLOFENAC SODIUM (SOLA Pharmaceuticals), DICLOFENAC SODIUM (Sandoz), DICLOFENAC SODIUM (Sportpharm), DICLOFENAC SODIUM (Sportpharm), DICLOFENAC SODIUM (St. Mary's Medical Park Pharmacy), DICLOFENAC SODIUM (St. Mary's Medical Park Pharmacy), DICLOFENAC SODIUM (St. Mary's Medical Park Pharmacy), DICLOFENAC SODIUM (Sun Pharmaceutical Industries), DICLOFENAC SODIUM (Sun Pharmaceutical Industries), DICLOFENAC SODIUM (Sun Pharmaceutical Industries), DICLOFENAC SODIUM (Umedica Laboratories USA), DICLOFENAC SODIUM 1.5% (PHOENIX RX LLC), DICLOFENAC SODIUM 1.5% (Proficient Rx LP), DICLOFENAC SODIUM 1.5% (SOLA Pharmaceuticals), DICLOFENAC SODIUM DELAYED RELEASE (A-S Medication Solutions), DICLOFENAC SODIUM DELAYED RELEASE (Cambridge Therapeutics Technologies), DICLOFENAC SODIUM GEL (Direct_Rx), DICLOFENAC SODIUM GEL (Direct_Rx), DICLOFENAC SODIUM GEL (Direct_Rx), DICLOFENAC SODIUM TOPICAL SOLUTION (Direct_Rx), DICLOFENAC SODIUM TOPICAL SOLUTION (Direct_Rx), VAROPHEN (V2 Pharma) |
|---|
| 組成式 |
C14H10Cl2NO2. Na
|
|---|
| 質量 |
316.9986
|
|---|
| 分子量 |
318.13
|
|---|
| 構造式 |
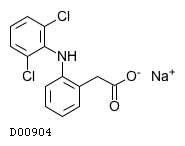
|
|---|
| Simcomp |
|
|---|
| クラス |
|
|---|
| コード |
| 商品 (DG00441): | D00903<US> D00904<JP/US> D07818<US> |
|
|---|
| 効能 |
鎮痛薬, 抗炎症薬, 解熱薬, シクロオキシゲナーゼ阻害薬 |
|---|
| ターゲット |
|
|---|
| パスウェイ |
|
|---|
| 代謝 |
Enzyme: CYP2C9 [HSA: 1559]
|
|---|
| 相互作用 |
Transporter inhibition: SLC22A8 [HSA: 9376]
|
|---|
| 構造マップ |
|
|---|
| 階層分類 |
医療用医薬品の薬効分類 [BR:jp08301]
1 神経系及び感覚器官用医薬品
11 中枢神経系用薬
114 解熱鎮痛消炎剤
1147 フェニル酢酸系製剤
D00904 ジクロフェナクナトリウム (JP18)
13 感覚器官用薬
131 眼科用剤
1319 その他の眼科用剤
D00904 ジクロフェナクナトリウム (JP18)
2 個々の器官系用医薬品
26 外皮用薬
264 鎮痛、鎮痒、収斂、消炎剤
2649 その他の鎮痛、鎮痒、収斂、消炎剤
D00904 ジクロフェナクナトリウム (JP18)
医療用医薬品のATC分類 [BR:jp08303]
D 皮膚科用薬
D11 その他の皮膚科用製剤
D11A その他の皮膚科用製剤
D11AX その他の皮膚科用製剤
D11AX18 ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18) <JP/US>
M 筋骨格系
M01 抗炎症及び抗リウマチ製剤
M01A 抗炎症及び抗リウマチ製剤、非ステロイド系
M01AB 酢酸誘導体と関連物質
M01AB05 ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18) <JP/US>
M02 関節・筋肉痛用局所薬剤
M02A 関節・筋肉痛用局所薬剤
M02AA 抗炎症製剤、局所用非ステロイド系
M02AA15 ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18) <JP/US>
S 感覚器
S01 眼科用薬
S01B 抗炎症薬
S01BC 抗炎症薬、非ステロイド性
S01BC03 ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18) <JP/US>
医療用医薬品のUSP分類 [BR:jp08302]
鎮痛薬
非ステロイド系抗炎症薬
ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18)
皮膚科用薬
皮膚科用薬, その他
イボ・日光角化症治療薬
ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18)
眼科用薬
眼科用抗炎症薬
非ステロイド系抗炎症薬, 眼科用薬
ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18)
一般用医薬品の分類 [BR:jp08313]
外皮用薬
57 鎮痛・鎮痒・収れん・消炎薬(パップ剤を含む)
D00904 ジクロフェナクナトリウム (JP18)
一般用医薬品のリスク区分 [BR:jp08312]
第二類医薬品
無機薬品及び有機薬品
ジクロフェナク
D00904 ジクロフェナクナトリウム (JP18)
医薬品グループ [BR:jp08330]
抗炎症薬
DG01504 非ステロイド性抗炎症薬 (NSAID)
DG01906 酢酸系抗炎症薬
DG01898 フェニル酢酸系抗炎症薬
DG00441 ジクロフェナク
D00904 ジクロフェナクナトリウム
代謝酵素基質薬
DG01642 CYP2C9基質薬
DG00441 ジクロフェナク
D00904 ジクロフェナクナトリウム
トランスポーター阻害薬
DG02866 SLC22A8阻害薬
DG00441 ジクロフェナク
D00904 ジクロフェナクナトリウム
医薬品クラス [BR:jp08332]
抗炎症薬
DG01504 非ステロイド性抗炎症薬 (NSAID) [商品一覧]
D00904 ジクロフェナクナトリウム
ターゲットに基づく医薬品分類 [BR:jp08310]
酵素
オキシドレダクターゼ (EC1)
プロスタグランジン合成酵素
PTGS1 (COX1)
D00904 ジクロフェナクナトリウム (JP18) <JP/US>
PTGS2 (COX2)
D00904 ジクロフェナクナトリウム (JP18) <JP/US>
日本薬局方収載医薬品 [BR:jp08311]
化学薬品等
D00904 ジクロフェナクナトリウム
D00904 ジクロフェナクナトリウム坐薬
薬物代謝酵素とトランスポーター [jp08309.html]
薬物代謝酵素
D00904
薬物トランスポーター
D00904
日本のスイッチOTC薬 [jp08314.html]
医療用から一般用へのスイッチOTC薬
D00904
医薬品グループ [BR:jp08330]
抗炎症薬
DG01504 非ステロイド性抗炎症薬 (NSAID)
DG01906 酢酸系抗炎症薬
DG01898 フェニル酢酸系抗炎症薬
DG00441 ジクロフェナク
代謝酵素基質薬
DG01642 CYP2C9基質薬
DG00441 ジクロフェナク
トランスポーター阻害薬
DG02866 SLC22A8阻害薬
DG00441 ジクロフェナク
|
|---|
| リンク |
|
|---|
| LinkDB |
|
|---|
|
» English version » Back
|